The Drug Administration of Vietnam – Ministry of Health has officially issued a document regarding the nationwide recall of a batch of Aceclofenac STELLA 100 mg film-coated tablets due to failure to meet quality standards. This is an urgent measure to ensure the safety of users and maintain strictness in pharmaceutical management in Vietnam.
Detailed information about the recalled drug batch
Based on current regulations, the drug batch suspended from circulation and recalled has the following specific identification information:
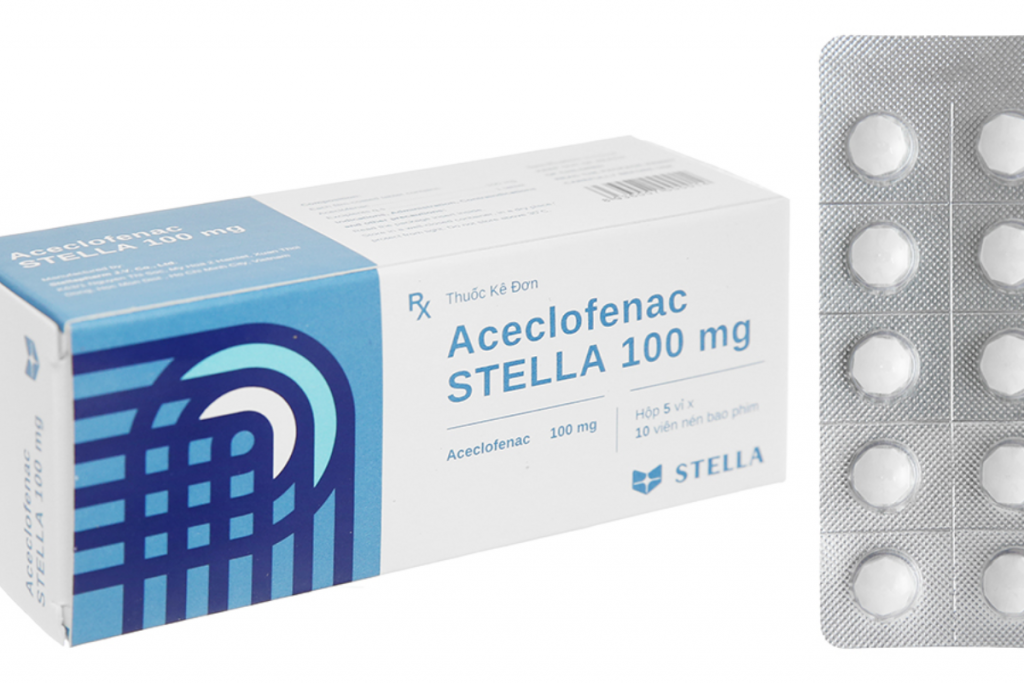
Aceclofenac STELLA 100 mg film-coated tablets
-
Drug name: Aceclofenac STELLA 100 mg film-coated tablets (Aceclofenac 100 mg).
-
Registration number: VD-20124-13.
-
Batch number: 070423.
-
Manufacturing date: 07/04/2023.
-
Expiration date: 07/04/2026.
-
Manufacturer: Stellapharm Joint Venture Company Limited.
Medical facilities and pharmaceutical businesses need to carefully cross-check the information on the packaging and the batch number to promptly classify and quarantine the products.
Causes and legal basis of the recall decision
The recall decision was issued after the Ho Chi Minh City Institute of Drug Quality Control sampled and tested the drug at Gia Phuc Pharmaceutical Company Limited. The test results indicated that batch number 070423 did not meet the quality standards regarding related impurities.
According to the classification by the functional agency, this is a level 2 quality violation. By regulation, level 2 violations include drug products with evidence of not fully ensuring treatment efficacy or posing an unsafe risk to users; however, this level has not yet caused serious harm to health or directly affected the lives of users at the time of detection.
Responsibilities of the manufacturer and distributor
Stellapharm Joint Venture Company Limited, as the manufacturer, is responsible for coordinating with relevant parties to strictly execute the recall procedure. Specifically, the enterprise must send recall notices to all wholesale and retail establishments, pharmacy chains, and medical examination and treatment facilities that have received the aforementioned drug batch.
The time limit to complete the organization of the recall and receive the violating drug volume is 15 days from the date the decision is issued. Furthermore, Stellapharm is responsible for covering all costs incurred during the recall process, handling the violating drugs, and compensating for damages in accordance with the provisions of the law. The recall result report must be compiled and submitted to the Drug Administration of Vietnam and the Ho Chi Minh City Department of Health within 03 days after concluding the recall work.
Guidelines for medical facilities and pharmaceutical businesses
To protect the rights and health of patients, the Drug Administration of Vietnam requires all pharmaceutical business establishments, including pharmacies and wholesale units, along with medical examination and treatment facilities nationwide, to implement the following actions:
-
Immediately cease the business, prescription, dispensing, and use of Aceclofenac STELLA 100 mg batch number 070423.
-
Return the goods to the supplier according to the manufacturer’s recall procedure.
-
Carefully inspect storage warehouses to avoid overlooking any products belonging to the violating batch.
Supervision and handling of violations by functional agencies
The Ho Chi Minh City Department of Health is assigned to directly inspect and supervise Stellapharm Joint Venture Company Limited in implementing the recall and handling of the substandard drug batch. Simultaneously, the Departments of Health of provinces and centrally-run cities are responsible for widely disseminating this information to medical facilities and pharmaceutical businesses within their jurisdictions.
Functional agencies will conduct unannounced inspections and supervise compliance with the recall order. Organizations and individuals that intentionally violate or fail to strictly implement the recall will be severely penalized according to the sanctions of pharmaceutical management laws.