Reorganization of the public health system: Focusing resources on leading hospitals
In the fourth week of March 2026, the healthcare sector recorded two prominent updates: A plan to reorganize the public health system to focus resources on leading hospitals and an urgent warning from the Drug Administration of Vietnam regarding the appearance of fake HIV pre-exposure prophylaxis drugs on the market.
The healthcare sector is entering a phase of strong restructuring of public service units, aiming for a lean, efficient system suitable for the increasing healthcare demand. This reorganization not only reduces the number of units but also restructures functions, tasks, decentralizes management, and allocates resources reasonably.
Prominent in this roadmap is the Ministry of Health’s merger of several units to enhance the connection between training and practice. Specifically, the National Hospital 71 and the National Rehabilitation Hospital will be merged into Hanoi Medical University. At the same time, the College of Medical Equipment Technology is merged into Viet Duc University Hospital, forming the Viet Duc College of Medicine and Medical Equipment.
Implementing Resolution No. 72 of the Politburo and Plan No. 130 of the Government Steering Committee, the Ministry of Health will gradually transfer some affiliated hospitals to local management. The Ministry of Health will only focus on managing specialized, high-tech, and leading hospitals to direct professional activities, train human resources, conduct scientific research, and respond to emergency situations. The reorganization ensures the principle of not interrupting medical examination and treatment activities, while helping hospitals expand their professional scope and improve service quality.
Warning on the discovery of fake YEZTUGO injection brand
Over the past week, the Drug Administration of Vietnam received reports of the sale and supply of fake HIV pre-exposure prophylaxis drugs counterfeiting the brand of Gilead Sciences Inc. The discovered drug sample is named YEZTUGO (lenacapavir) injection, with the manufacturing location listed as the US, but it completely lacks a valid registration number. Search results on the public service system show that the Drug Administration of Vietnam has never issued a circulation registration certificate for this product or a manufacturing facility with the above name in the Vietnamese market.
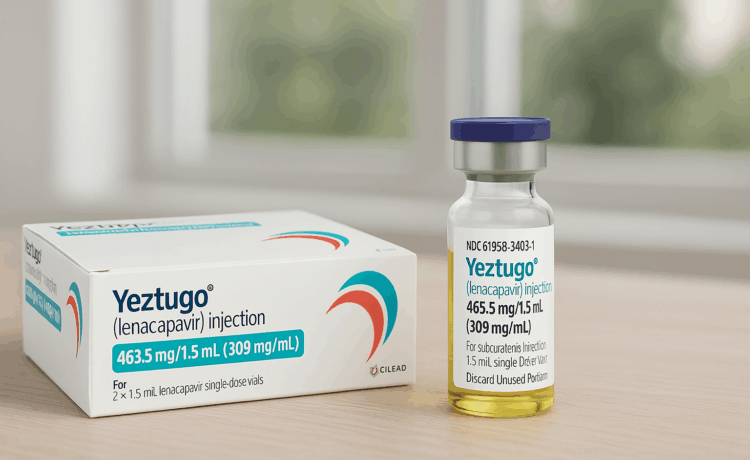
Yeztugo injection
To protect patient health, the Drug Administration of Vietnam has requested the Ho Chi Minh City Department of Health to continue verifying and strictly handling the case. At the same time, the Departments of Health of provinces and centrally-run cities need to quickly notify business establishments, medical examination and treatment facilities, and citizens to thoroughly prevent the trading and use of the unlicensed YEZTUGO injection product.
Pharmacologically, YEZTUGO injection is the trade name of the active ingredient lenacapavir, belonging to the class of HIV antiviral drugs, playing an important role in pre-exposure prophylaxis. The use of fake, undocumented drugs containing this active ingredient will cause serious consequences, increasing the risk of exposure and reducing the effectiveness of national HIV/AIDS prevention programs.
Center for Support & Continuing Education